|
Thus, we hypothesize that the inhibition of (net) oxidative dissolution at pH 2 may be the result of secondary precipitation of a chromic hydroxy chromate phase. However, sorption of Cr(VI) (putatively formed during oxidative dissolution) cannot explain the observed inhibition of the reaction because (1) sorption occurs at both pH 2 and 3 but inhibition only at pH 2 and (2) preequilibration of Cr(OH)3(S) with Cr(VI) did not affect the rate of dissolution observed upon the addition of the oxidant. Cr(VI) sorption followed a Langmuir isotherm and reached maximum sorption densities of 308 ± 8 and 271 ± 10 μmol/g at pH 3 and 2, respectively. Under oxidizing conditions, Cr released into solution is expected to be present as Cr(VI), which sorbs strongly to Cr(OH)3(S) at low pH. We established that the plain Cr (III) hydroxide, abiding to the approximate molecular formula Cr (OH) 3 ♳H 2 O, was crystalline, highly soluble, i.e. In contrast, the rate of Cr release at pH 2 decreased rapidly with time, and within 2.5 h, the dissolution reaction was completely inhibited. Linear kinetics of oxidative dissolution was observed at pH 9 and 3. Chromic Cation Cr+8 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety. 200 and to a lesser extent at pH 2 and 3. 
The skeleton ionic reaction is Cr(OH) (s) + IO3 (aq) CrO(aq) + I¯(aq) Definition Definition Chemical reactions involving both oxidation and reduction processes. 
If instead a strong acid is added to the solution, the added H + will react essentially completely with A to form HA. Dissolution of Cr(OH)3(S) (1.0 g/L) was accelerated in the presence of excess strong oxidant (20 mM) at pH 9 by a factor of ca. Transcribed Image Text: Write a balanced molecular equation for the reaction between chromium(II) hydroxide and sodium iodate in basic solution. Because of the reaction described in Equation 18.7.1 18.7.1, the predicted solubility of a sparingly soluble salt that has a basic anion such as S 2, PO 43, or CO 32 is increased. Dissolution of Cr(OH)3(S) in 0.01 M NaNO3 suspensions was examined in batch experiments in the presence and absence of the strong oxidant sodium hypochlorite (NaOCl). General Chemistry - Standalone book (MindTap Course List) 11th Edition. Use this information to calculate a Ksp value for chromium(III) hydroxide. This Thermo Scientific Chemicals brand product was originally.44. 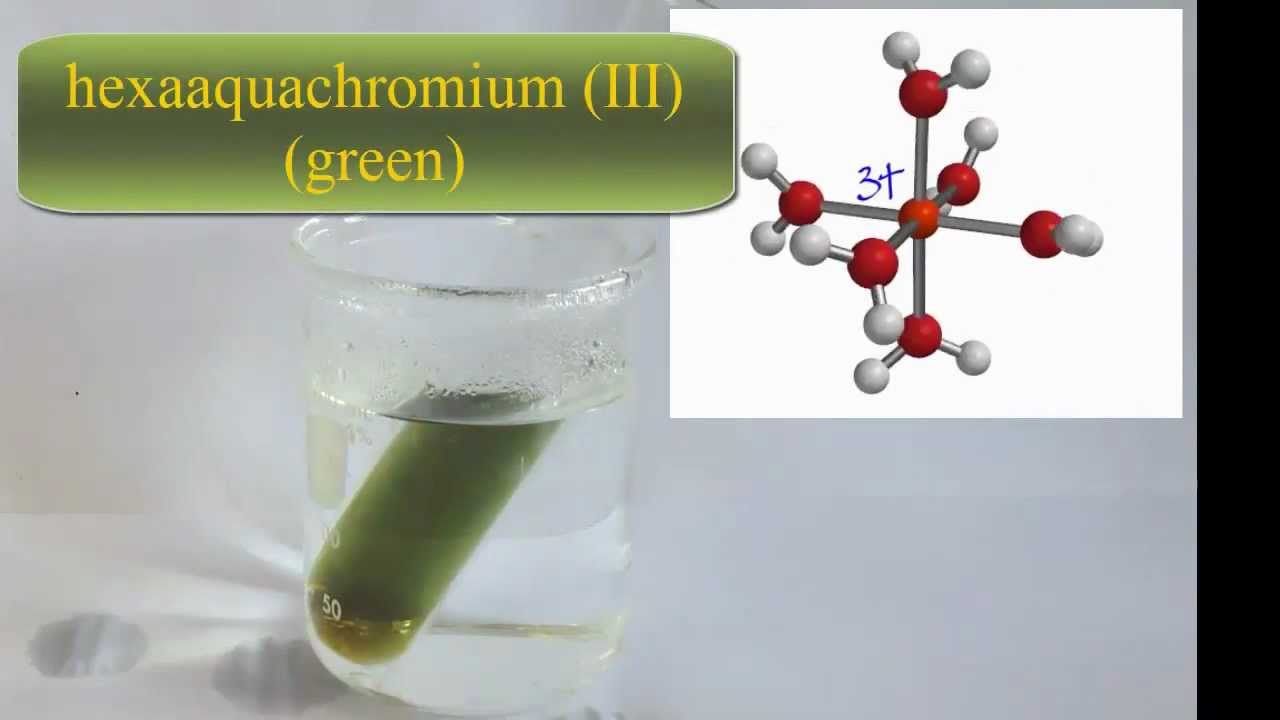
used as an oxygen scrubber, polymer industry. Conversely, the oxidation of Cr(III)to Cr(VI) increases both its toxicity and often its mobility. The solubility of Cr(OH)3 is measured and found to be 1.31x10-6 g/L. Occasionally to dehalogenate organic compounds. Hexavalent chromium, Cr(VI), can be immobilized under neutral to alkaline conditions by reduction to Cr(III) similarly, the mobility of naturally occurring Cr in soils and sediments can be limited by its occurrence in the +III oxidation state.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
